Introduction
Pediatric and young adult patients with primary intracranial tumors often require multimodal therapy that may include surgery, chemotherapy, and Radiotherapy (RT). Although advances such as Intensity-Modulated Radiotherapy (IMRT) have improved dose conformity and reduced exposure to surrounding normal tissues, the hypothalamic–pituitary axis remains vulnerable due to its close anatomical relationship with many Central Nervous System (CNS) tumor sites. Even limited radiation-related injury to this region can result in clinically meaningful hormonal disturbances because of its central role in regulating growth, metabolism, reproductive function, and stress response.
Among the potential late effects of cranial irradiation, thyroid axis dysfunction is one of the more frequently observed endocrine changes. Presentation may be subtle and develop gradually, with symptoms such as fatigue, slowed growth, altered metabolic activity, or cognitive effects. These features can be easily overlooked without structured follow-up. The likelihood and severity of endocrine disruption are influenced by several factors, including total radiation dose, fractionation schedule, patient age at the time of exposure, and proximity of the tumor and target volume to the hypothalamic–pituitary region.
Reported incidence rates of radiation-induced hypothyroidism vary widely across studies, partly due to differences in radiation techniques, follow-up duration, and hormonal monitoring strategies. Although IMRT allows for improved dose sculpting, incidental radiation to the pituitary gland may remain unavoidable in tumors near the sellar, suprasellar, or posterior fossa regions.
This study was undertaken to evaluate the incidence, pattern, and timing of hypopituitarism, with particular emphasis on thyroid axis dysfunction, in pediatric and young adult patients treated with radiotherapy for intracranial tumors. We also assessed whether these outcomes correlated with the mean radiation dose delivered to the pituitary gland.
Materials and methods
Study design and participants
This was a prospective observational study conducted between January 2015 and December 2018. A total of 30 patients aged 6–25 years with histologically confirmed primary intracranial tumors who received adjuvant or radical radiotherapy were included. Patients with pre-existing thyroid or pituitary disease were excluded.
Ethical considerations
This study involved minimal-risk observational monitoring and did not require formal institutional ethical committee approval. Informed consent was obtained from patients or guardians, and the study adhered to the principles of the Declaration of Helsinki.
Radiotherapy technique
All patients underwent treatment planning using CT-based simulation with MRI fusion. Target volumes were contoured per standard CNS tumor guidelines. Treatment was delivered using IMRT with dose prescriptions according to tumor histology and clinical status. The pituitary gland and hypothalamus were contoured as organs at risk. Mean pituitary dose was recorded for analysis.
Hormonal evaluation
Baseline assessments of TSH, free T3 (FT3), free T4 (FT4), LH, FSH, GH, ACTH, and prolactin were obtained prior to RT. Follow-up testing was performed at 3, 6, and 9 months post-treatment. Hypothyroidism was defined as elevated TSH with low FT4; subclinical hypothyroidism was defined as elevated TSH with normal FT4.
Results
The mean age of the study population was 27.32±2.84 years. The most common tumor types were anaplastic astrocytoma and medulloblastoma, each accounting for 28% of cases.
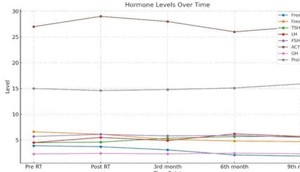
Table 1: Serial changes in pituitary hormone levels before and
after radiotherapy.
| Hormone |
Pre-RT (mean ± SD) |
Post-RT (mean ± SD) |
3rd Month |
6th Month |
9th Month |
| Free T3 |
3.9±0.3 |
3.7±0.3 |
3.1±0.3 |
2.1±0.2 |
1.8±0.2 |
| Free T4 |
6.6±0.6 |
6.1±0.5 |
5.1±0.4 |
4.8±0.4 |
4.7±0.3 |
| TSH |
4.5±0.4 |
4.6±0.4 |
5.3±1.5 |
5.7±1.5 |
5.7±1.5 |
| LH |
4.5±0.3 |
5.5±0.5 |
4.9±0.6 |
6.2±1.3 |
5.7±1.2 |
| FSH |
5.7±1.3 |
6.1±1.3 |
5.8±1.2 |
5.9±1.2 |
5.5±1.1 |
| ACTH |
27±1.3 |
29±13.1 |
28±12.9 |
26±12.1 |
27±12.8 |
| GH |
2.3±0.6 |
2.4±0.7 |
2.3±0.5 |
2.45±0.4 |
2.34±0.5 |
| Prolactin |
15±2.3 |
14.6±2.1 |
14.8±2.3 |
15.1±2.1 |
15.9±2.2 |
Values are expressed as mean ± standard deviation. Values marked with: *P<0.05 compared to Pre‑RT; **p<0.01 compared to Pre‑RT
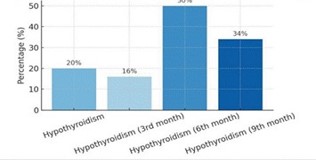
Thyroid axis function
Six patients (20%) developed hypothyroidism during follow-up. One case was detected at 3 months post-RT, three cases at 6 months, and two cases at 9 months. Subclinical hypothyroidism occurred in an additional two patients. All patients with thyroid dysfunction had a mean pituitary dose greater than 40 Gy.
Other pituitary hormones
LH, FSH, GH, ACTH, and prolactin levels remained within normal limits across all follow-up intervals, with no clinical evidence of secondary adrenal or gonadal dysfunction.
Discussion
In this study, thyroid axis dysfunction was observed in 20% of pediatric and young adult patients following radiotherapy for intracranial tumors. The majority of cases were identified within 6 to 9 months after treatment, indicating that thyroid hormone alterations may develop earlier in the post-treatment period than is generally anticipated. This highlights the importance of structured endocrine follow-up within the first year after radiotherapy, rather than delaying assessment until later survivorship.
The association between hypothyroidism and mean pituitary doses exceeding 40 Gy in our cohort aligns with the dose-response relationship described in previous literature. Although IMRT improves dose conformity around critical structures, complete avoidance of the hypothalamic–pituitary axis may be challenging when treating tumors in the sellar, suprasellar, midline, or posterior fossa regions. Incorporating pituitary-sparing dose constraints during planning and reviewing dose-volume parameters routinely may help reduce the likelihood of delayed endocrine effects.
In contrast to several reports identifying growth hormone deficiency as one of the earliest manifestations of radiation-related pituitary injury, no significant abnormalities in GH, LH, FSH, ACTH, or prolactin levels were observed during the follow-up period in this study. This may reflect the relatively short surveillance duration, as deficiencies in these hormonal axes can emerge gradually over several years. Continued longitudinal monitoring remains essential to detect delayed dysfunction.
Early recognition and management of hypothyroidism are particularly important in younger patients, as untreated thyroid deficiency can affect growth velocity, cognitive performance, mood, energy levels, and overall quality of life. Integrating endocrine evaluation into routine post-treatment care can support timely intervention and mitigate long-term morbidity.
Advances in radiotherapy planning, including adaptive planning and the use of proton beam therapy where available, may further reduce radiation exposure to the hypothalamic–pituitary axis. Multidisciplinary coordination between oncologists, endocrinologists, and survivorship care teams is key to optimizing long-term outcomes in this patient population.
Conclusion
Radiotherapy for CNS tumors in pediatric and young adult patients is associated with a measurable risk of thyroid axis dysfunction, particularly when the mean pituitary dose exceeds 40 Gy. Early and periodic assessment of thyroid function is recommended to facilitate timely management. Efforts to minimize pituitary dose during radiotherapy planning should be prioritized to reduce late endocrine morbidity.
• Radiation-induced hypothyroidism occurred in 20% of pediatric and young adult patients treated for primary intracranial tumors.
• Most thyroid dysfunction developed early, within 6–9 months after completing radiotherapy.
• All cases of hypothyroidism were associated with a mean pituitary dose exceeding 40 Gy, demonstrating a clear dose–response relationship.
• Other pituitary hormonal axes (LH, FSH, GH, ACTH, prolactin) remained preserved during short-term follow-up.
• Findings emphasize the need for routine early endocrine monitoring and incorporation of pituitary-sparing radiotherapy techniques to reduce long-term morbidity.
Declarations
Conflict of interest statement: The authors declare that there are no conflicts of interest regarding the publication of this manuscript.
Funding statement: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for profit sectors.
Ethics statement: This study involved minimal-risk observational data collection. Formal institutional ethics committee approval was not required, and the study adhered to the principles of the Declaration of Helsinki.
Written informed consent was obtained from all participants or their legal guardians.
Data availability statement: The data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
- Littley MD, Shalet SM, Morgenstern GR, et al. Endocrine and reproductive dysfunction following fractionated total body irradiation in adults. Q J Med. 1991; 78: 265–274.
- Srikantia N, Karthik S, Mala K. How common is hypothyroidism after external radiotherapy to head and neck cancers? Indian J Med Paediatr Oncol. 2011; 32: 143–148.
- Darzy KH. Radiation-induced hypopituitarism after cancer therapy: who, how and when to test. Nat Clin Pract Endocrinol Metab. 2009; 5: 88–99.
- Merchant TE, Li C, Xiong X, et al. Conformal radiotherapy after surgery for pediatric ependymoma: a prospective study. Lancet Oncol. 2008; 9: 573–580.
- Constine LS, Woolf PD, Cann D, et al. Hypothalamic–pituitary dysfunction after radiation for brain tumors. Int J Radiat Oncol Biol Phys. 1993; 27: 493–502.
- Shalet SM, Shoubridge EA, Morris-Jones PH. Effects of radiation therapy on growth hormone secretion in childhood. Endocrinol Metab Clin North Am. 1996; 25: 575–590.
- Darzy KH, Shalet SM. Hypopituitarism following radiotherapy. Horm Res. 2005; 64: 1–6.
- Samaan S, Alsaad AA, Ismail HM, et al. Central hypothyroidism in survivors of pediatric brain tumors: prevalence, risk factors and longterm outcomes. J Clin Endocrinol Metab. 2021; 106: 2693–2704.
- Patterson BC, Bilodeau M, Kelly KM, et al. Endocrine outcomes following proton and photon radiotherapy for pediatric brain tumors. Pediatr Blood Cancer. 2022; 69: e29430.
- Cohen LE. Endocrine late effects of cancer treatment. Endocrinol Metab Clin North Am. 2020; 49: 569–587.
- Armstrong GT, Stovall M, Robison LL. Long-term effects of radiation exposure among adult survivors of childhood cancer: results from the Childhood Cancer Survivor Study. Radiat Res. 2010; 174: 840– 850.
- Thomas BC, Gan GN, Armstrong GT, et al. Reduced hypothalamic–pituitary axis risk with proton therapy compared to photon therapy in childhood CNS tumors: a systematic review. Neuro Oncol. 2023; 25: 251–263.